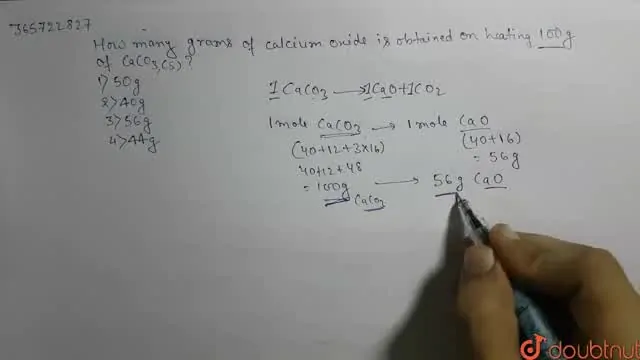
How many grams of calcium oxide is obtained on heating 100 g of CaCO3(s)?
50 g
40 g
56 g
44 g
The correct Answer is:C
Step by step video, text & image solution for How many grams of calcium oxide is obtained on heating 100 g of CaCO_(3)(s)? by Chemistry experts to help you in doubts & scoring excellent marks in Class 12 exams.
Related Playlists
- BOOK - AAKASH INSTITUTECHAPTER - SOME BASIC CONCEPT OF CHEMISTRYEXERCISE - ASSIGNMENT( SECTION - A) Objective Type Questions20videos
SOME BASIC CONCEPT OF CHEMISTRY
- BOOK - AAKASH INSTITUTECHAPTER - SOME BASIC CONCEPT OF CHEMISTRYEXERCISE - ASSIGNMENT( SECTION - B) Objective Type Questions22videos
SOME BASIC CONCEPT OF CHEMISTRY
- BOOK - AAKASH INSTITUTECHAPTER - SOME BASIC CONCEPT OF CHEMISTRYEXERCISE - TRY YOURSELF30videos
SOME BASIC CONCEPT OF CHEMISTRY
- BOOK - AAKASH INSTITUTECHAPTER - SOLUTIONSEXERCISE - ASSIGMENT (SECTION-J) AAKASH CHALLENGERS QUESTIONS10videos
SOLUTIONS
- BOOK - AAKASH INSTITUTECHAPTER - SOME BASIC CONCEPTS OF CHEMISTRY EXERCISE - ASSIGNMENT SECTION J (Aakash Challenges Questions)10videos
SOME BASIC CONCEPTS OF CHEMISTRY
Similar Questions
Calcium carbide is obtained by heating
View SolutionWhat mass of calcium oxide will be obtained by heating 3 mol of CaCO3 ?
View SolutionCalcium carbonate decomposes on heating according to the following equations:
CaCO3(s)⇔CaO(s)+CO2(g)
How many moles of CO2 will be obtained by decomposition of 50g of CaCO3?View SolutionThe molar mass of CaCO3 is 100 g. The maximum amount of carbon dioxide that can be liberated on heating 25 g of CaCO3 is -
View SolutionHow many atoms of each constituent element are present in 50 g of CaCO3 ?
View SolutionThe volume of CO2 at STP obtained by heating 1 g of CaCO3 will be :
View SolutionThe mass of CaO obtained by heating 100 kg of 95% pure limestone (CaCO3) is-
View SolutionThe volume of CO2 evolved at STP on heating 50g CaCO3
View SolutionFind the volume of CO2 obtained at S.T.P. on heating 200 gm of 50% pure CaCO3 -
View SolutionWhat mass of CaO is formed by heating 50 g CaCO3 in air?
View Solution